VABOMERE® dosing in patients 18 years of age and older1
DOSING IN PATIENTS WITH cUTI1

Recommended dosage1:
4 grams* administered every 8 hours by intravenous (IV) infusion over 3 hours in patients ≥18 years of age with estimated glomerular filtration rate (eGFR) ≥50 mL/min/1.73 m2
The duration of treatment can be up to 14 days
4 g of VABOMERE (meropenem and vaborbactam) = 2 g meropenem and 2 g of vaborbactam.
Dosage adjustment is recommended in patients with renal impairment who have an eGFR <50 mL/min/1.73 m2.
DOSING IN PATIENTS WITH RENAL IMPAIRMENT1

*As calculated using the Modification of Diet in Renal Disease (MDRD) formula as follows: eGFR (mL/min/1.73m2) =175 x (serum creatinine)-1.154 x (age)-0.203 x (0.742 if female) x (1.212 if African American).
†All doses of VABOMERE are administered IV over 3 hours.
‡Doses adjusted for renal impairment should be administered after a hemodialysis session.
§The total duration of treatment is for up to 14 days.
- Meropenem and vaborbactam can be removed by hemodialysis1
- For patients maintained on hemodialysis, VABOMERE should be administered after a hemodialysis session1
- For patients with changing renal function, monitor serum creatinine concentrations and eGFR at least daily and adjust the dosage of VABOMERE accordingly1
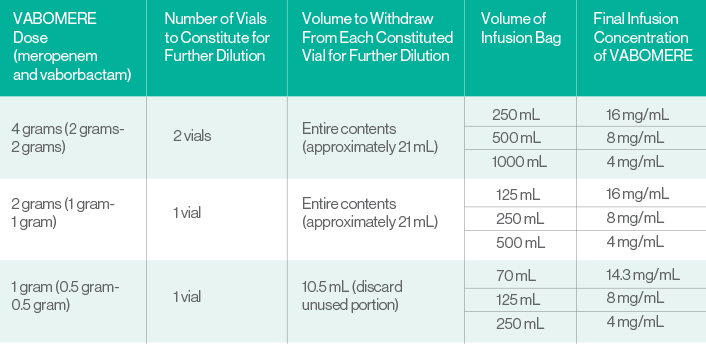
Preparation and administration of VABOMERE for intravenous infusion1
VABOMERE is supplied as a dry powder in a single-dose vial that must be constituted and further diluted using aseptic technique prior to intravenous infusion as outlined below. VABOMERE does not contain preservatives.
- Prepare the required dose by constituting the appropriate number of vials (see Table below). Withdraw 20 mL of 0.9% Sodium Chloride Injection, USP from an infusion bag and constitute each vial of VABOMERE.
- Mix gently to dissolve. The constituted VABOMERE solution is not for direct injection.
- Immediately dilute the constituted solution further by withdrawing full or partial constituted vial contents from each vial and add back into the 0.9% Sodium Chloride Injection, USP infusion bag (see Table).
- Complete the intravenous infusion of the diluted solution within 4 hours if stored at room temperature or 22 hours if stored refrigerated at
2°C to 8°C (36°F to 46°F).
Prior to administration:
Visually inspect the diluted VABOMERE solution for particulate matter and discoloration (the color of the VABOMERE infusion solution for administration ranges from colorless to light yellow). Discard unused portion after use.
PREPARATION OF VABOMERE DOSES1
Reference: 1. VABOMERE [package insert]: Melinta Therapeutics, LLC.
INDICATION AND USAGE
VABOMERE® (meropenem and vaborbactam) is indicated for the treatment of patients 18 years of age and older with complicated urinary tract infections (cUTI) including pyelonephritis caused by the following susceptible microorganisms: Escherichia coli, Klebsiella pneumoniae, and Enterobacter cloacae species complex.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of VABOMERE® and other antibacterial drugs, VABOMERE® should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria.
IMPORTANT SAFETY INFORMATION
Contraindications
VABOMERE® is contraindicated in patients with known hypersensitivity to any components of VABOMERE® (meropenem and vaborbactam), or to other drugs in the same class or in patients who have demonstrated anaphylactic reactions to beta-lactam antibacterial drugs.
Warnings and Precautions
- Hypersensitivity reactions were reported in patients treated with VABOMERE® in the clinical trials. Serious and occasionally fatal hypersensitivity (anaphylactic) reactions and serious skin reactions have been reported in patients receiving therapy with beta-lactam antibacterial drugs. There have been reports of individuals with a history of penicillin hypersensitivity who have experienced severe hypersensitivity reactions when treated with another beta-lactam antibacterial drug. If an allergic reaction to VABOMERE® occurs, discontinue the drug immediately.
- Seizures and other adverse Central Nervous System (CNS) experiences have been reported during treatment with meropenem, which is a component of VABOMERE®. Close adherence to the recommended dosage regimens is urged, especially in patients with known factors that predispose to convulsive activity.
- Rhabdomyolysis has been reported with the use of meropenem, a component of VABOMERE® If signs or symptoms of rhabdomyolysis such as muscle pain, tenderness or weakness, dark urine, or elevated creatine phosphokinase are observed, discontinue VABOMERE and initiate appropriate therapy.
- Clostridioides difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including VABOMERE®, and may range in severity from mild diarrhea to fatal colitis. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents. If CDAD is suspected or confirmed, ongoing antibacterial drug use not directed against C. difficile may need to be discontinued.
- The concomitant use of VABOMERE® and valproic acid or divalproex sodium is generally not recommended. Case reports in the literature have shown that co-administration of carbapenems, including meropenem, to patients receiving valproic acid or divalproex sodium results in a reduction in valproic acid concentrations. The valproic acid concentrations may drop below the therapeutic range as a result of this interaction, therefore increasing the risk of breakthrough seizures. If administration of VABOMERE® is necessary, consider supplemental anticonvulsant therapy.
- In patients with renal impairment, thrombocytopenia has been observed in patients treated with meropenem, but no clinical bleeding has been reported.
- Alert patients receiving VABOMERE® on an outpatient basis regarding adverse reactions such as seizures, delirium, headaches and/or paresthesias that could interfere with mental alertness and/or cause motor impairment.
- Prescribing VABOMERE® in the absence of a proven or strongly suspected bacterial infection is unlikely to provide benefit to the patient and increases the risk of drug-resistant bacteria.
- As with other antibacterial drugs, prolonged use of VABOMERE® may result in overgrowth of nonsusceptible organisms.
Adverse Reactions
The most frequently reported adverse reactions occurring in ≥3% of patients treated with VABOMERE® were headache, phlebitis/infusion site reactions, and diarrhea.
Please see full Prescribing Information.|
Distribution Change for AmerisourceBergen, Cardinal, and McKesson – Now Order Exclusively through Specialty Distribution |
x
|