VABOMERE®: A combination of meropenem and vaborbactam designed to address resistant Enterobacterales1
VABOMERE® (meropenem and vaborbactam) is specifically designed to restore the power of meropenem against KPC-producing Enterobacterales1
VABOMERE combines meropenem, a trusted carbapenem, with vaborbactam, a unique β-lactamase inhibitor1*
VABOMERE maintains the well established safety profile of meropenem1-3
Common adverse reactions for VABOMERE in the TANGO I trial were consistent with those reported for meropenem alone1-3
VABOMERE demonstrates activity against a broad spectrum of gram-negative pathogens1
In vitro activity against Enterobacter cloacae species complex, E coli, K pneumoniae, P aeruginosa, and ESBLs1*
VABOMERE demonstrated a greater overall success rate in a clinical trial than a widely used cUTI treatment1,3†
98.4% with VABOMERE vs 94.3% with piperacillin/tazobactam1
Infectious Diseases Society of America (IDSA) recommendations for the use of VABOMERE4
Recommendations from 2024 IDSA Guidance, CRE section, Questions 3.2, 3.3, and 3.4
- VABOMERE® (meropenem and vaborbactam) is a preferred treatment option for cUTI or pyelonephritis caused by CRE
- VABOMERE is also a preferred treatment option for infections caused by CRE that are not carbapenemase producing and that do not exhibit susceptibility to any carbapenem
- VABOMERE is slightly favored for KPC-producing infections followed by ceftazidime-avibactam and imipenem-cilastatin-relebactam
- Guidance is based on available data regarding clinical outcomes and emergence of resistance
- Clinical cure and 30-day mortality rates between patients who received VABOMERE and patients who received ceftazidime-avibactam numerically favored VABOMERE in an observational study
- IDSA suggests reserving cefiderocol for the treatment of infections caused by metallo-β-lactamase-producing Enterobacterales or glucose non-fermenting gram-negative organisms
Emergence of resistance to novel β-lactam antibiotics4
While the emergence of resistance is a concern with all of the novel β-lactams used to treat CRE infections, IDSA notes that the frequency may be highest for ceftazidime-avibactam.
with VABOMERE
with ceftazidime-
avibactam
IDSA recommends always repeating susceptibility testing for patients previously infected with CRE who present with symptoms suggestive of a new or relapsed infection. Patients recently treated with ceftazidime-avibactam may be treated with a different novel β-lactam agent such as VABOMERE at least until culture and susceptibility data are available.
Development of clinical resistance in real-world use
In support of its findings, IDSA discussed a single observational study comparing the clinical outcomes of 26 patients who received VABOMERE and 105 patients who received ceftazidime-avibactam for at least 72 hours for the treatment of CRE infections.
Percentage of patients with recurrent CRE infections who developed resistance to initial therapy:
0% with VABOMERE (n= 0/3) vs. 20% with ceftazidime-avibactam (n=3/15)
These statements are not intended to imply comparable safety or effectiveness between VABOMERE and ceftazidime-avibactam. Consult the respective products’ Prescribing Information for further details, including complete indication and Important Safety Information. Observational studies contain material limitations and their results should be considered in light of the entire body of available evidence, including clinical trial data.
cUTIs due to gram-negative bacteria are increasing mainly because of ESBL-producing Enterobacterales5
The most common causes of cUTI are gram-negative bacteria5
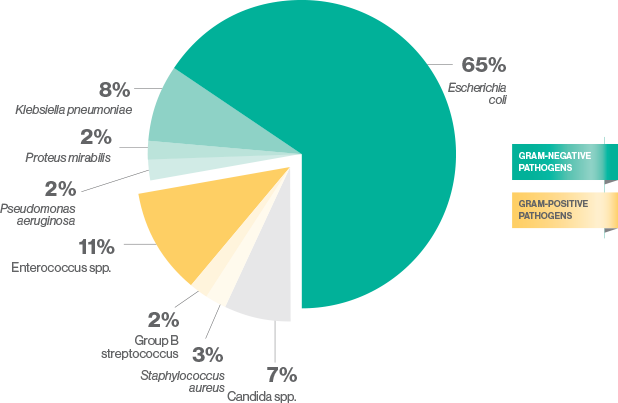
ESBL=extended spectrum ß-lactamase.
- Compared to younger patients, elderly patients are more likely to have a cUTI due to K pneumoniae or P aeruginosa6
Antibiotic resistance is a serious threat in the US7
Estimated numbers of infections and deaths caused by antibiotic resistance*:
>2.8 million
infections
>35,000
deaths
*Includes bacterial and fungal infections.
The 2019 CDC Antibiotic Resistance Threats in the United States report includes 18 antibiotic-resistant bacteria and fungi—classified as serious, urgent, or concerning. The report classifies carbapenem-resistant Enterobacterales (CRE) as urgent and ESBL-producing Enterobacterales as serious.7
KPC-producing CRE are prevalent in the US8,9
KPC was the most common gene detected among CRE isolates8

42,006 CRE isolates were tested from January 2017 through December 2019 by the Antibiotic Resistance Lab Network8
KPC-producing CRE have been identified in every state9

CRE—an urgent threat to your most vulnerable patients10
CRE infections are significantly more likely in patients with the following risk factors:
Chronic moderate-to-severe renal insufficiency3,10,11
≥3 comorbidities3,10
Prior CRE
infection10,11
Immune
compromise10-12
Prolonged hospitalization or antibiotic therapy10-13
Indwelling
catheters3,10,12
Long-term care
in a nursing facility11,12
High-risk patients frequently experience poor outcomes10
Vulnerable patients experience lower rates of clinical cure and higher rates of 28-day mortality.10
Delaying appropriate treatment of CRE infections can contribute to poor outcomes among critically ill patients.14,15
VABOMERE patient profiles
Xavier, 78 yrs

Assisted living resident with
indwelling urinary catheter,
admitted to ICU with elevated fever and
an altered mental state from baseline related to suspected cUTI
Assisted living resident with indwelling urinary catheter, admitted to ICU with elevated fever and an altered mental state from baseline related to suspected cUTI
Kathy, 62 yrs

History of hospitalizations for cUTI and multiple comorbidities
including renal impairment
admitted to ICU admitted to the ICU for suspected cUTI
History of hospitalizations for cUTI and multiple comorbidities, including renal impairment, admitted to ICU admitted to the ICU for suspected cUTI
Donna, 70 yrs

Immunocompromised due to
ongoing immunotherapy and
chemotherapy, history of CRE
infection, admitted to ICU
for suspected cUTI
Immunocompromised due to ongoing immunotherapy and chemotherapy, history of CRE infection, admitted to ICU for suspected cUTI
Martin, 68 yrs

Admitted to ICU post
CABG with suspected cUTI
2 days post-urinary
catheter removal,
multiple comorbid conditions
Admitted to ICU post CABG with suspected cUTI 2 days post-urinary catheter removal, multiple comorbid conditions
Timothy, 81 yrs

Nursing home resident
with indwelling catheter,
admitted to ICU with elevated
fever and confusion believed
to be related to cUTI
Nursing home resident with indwelling catheter, admitted to ICU with elevated fever and confusion believed to be related to cUTI
These hypothetical case studies are meant to be illustrative. They
are not intended to offer medical advice. Determination of
appropriate treatment is at the discretion of the physician. Treatment results may vary by
patient.









