The efficacy of VABOMERE® was proven against a widely used anti-infective agent1,2
VABOMERE® (meropenem and vaborbactam) demonstrated an overall success rate of 98.4% vs 94.3% with piperacillin/tazobactam1-3
- Overall success at the end of intravenous treatment (EOIVT) included clinical cure or improvement and microbiological eradication1,2*†
- Clinical cure and microbiological eradication was assessed at the test of cure (TOC) visit approximately 7 days after completion of treatment1‡§
CLINICAL AND MICROBIOLOGICAL RESPONSE RATES (m-MITT)
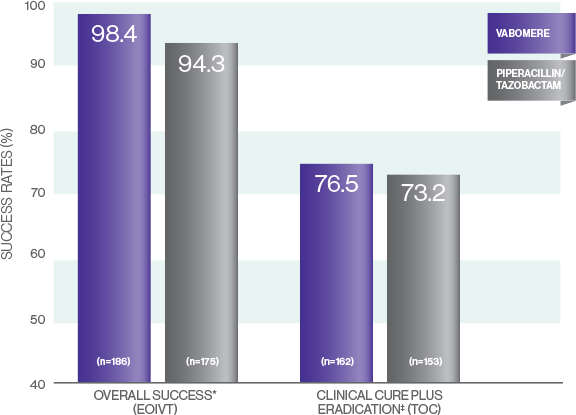
*EOIVT includes patients with organisms resistant to piperacillin/tazobactam at baseline.2,3
†Primary endpoint: 4.1% treatment difference (95% CI, 0.3%-8.8%) exceeded the lower limit for both noninferiority and superiority.2,3
‡TOC visit excludes patients with organisms resistant to piperacillin/tazobactam at baseline in both arms.2,3
§3.3% treatment difference (95% CI, -6.2%-13%).2,3
TANGO I Study description
A double-blind, double-dummy, randomized, multicenter noninferiority clinical trial evaluated 545
adult patients with cUTI, including acute pyelonephritis. Patients were treated with VABOMERE
(meropenem 2 g/vaborbactam 2 g infused over 3 hours) or piperacillin/tazobactam (piperacillin 4
g/tazobactam 0.5 g infused over 30 mintues) every 8 hours. After a minimum of 15 doses of IV therapy,
patients who met prespecified criteria of improvement could be switched to oral levofloxacin.1,2
Primary endpoint: Overall success at the EOIVT (composite of both a clinical outcome of cure or
improvement and a microbiologic outcome of eradication) in the m-MITT population.1,2
Suspect a serious gram-negative cUTI? Defend your at-risk patients with VABOMERE.
Proven safety and tolerability comparable to meropenem1,2,4
ADVERSE REACTIONS OCCURRING IN 1% OR GREATER OF PATIENTS
RECEIVING VABOMERE IN THE PHASE 3 CLINICAL TRIAL IN cUTI1,2
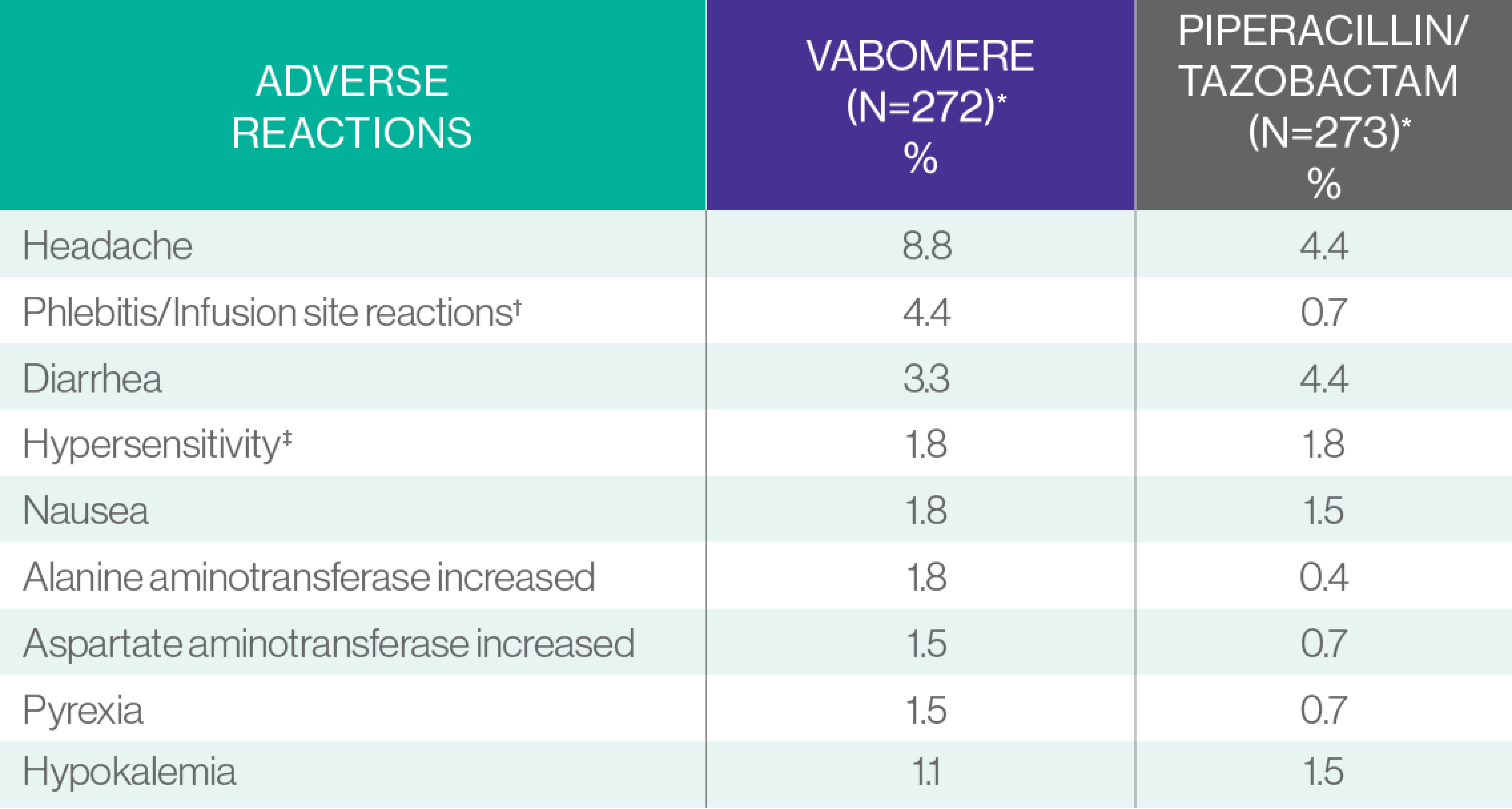
*Piperacillin/tazobactam 4.5 g (piperacillin 4 g/tazobactam 0.5 g) IV infused over 30 minutes every 8 hours. VABOMERE 4 g (meropenem 2g/vaborbactam 2 g) IV infused over 3 hours every 8 hours.
†Infusion site reactions include infusion/injection site phlebitis, infusion site thrombosis, and infusion site erythema.
‡Hypersensitivity includes hypersensitivity, drug hypersensitivity, anaphylactic reaction, rash urticaria, and bronchospasm.
The majority of adverse events with VABOMERE were either mild or moderate in severity.1
Fewer discontinuations due to adverse reactions with VABOMERE than with piperacillin/tazobactam1
- Adverse reactions leading to discontinuation were reported in 2.9% (8/272) vs 5.1% (14/273) of patients receiving VABOMERE vs patients receiving piperacillin/tazobactam, respectively1
- The most frequently reported adverse reactions leading to discontinuation of VABOMERE were hypersensitivity (1.1%; 3/272) and infusion‑related reactions (0.7%; 2/272)1
VABOMERE maintains the well‑established safety profile of meropenem1,2,4
- The most common adverse reactions for VABOMERE, as seen in the Phase 3 clinical trials, were consistent with those reported for meropenem alone1,2,4
Study description
A double-blind, double dummy, randomized, multicenter noninferiority clinical trial evaluated 545 adult
patients with cUTI, including acute pyelonephritis. Patients were treated with VABOMERE (meropenem 2
g-vaborbactam 2 g infused over 3 hours) or piperacillin/tazobactam (piperacillin 4 g/tazobactam 0.5 g
infused over 30 mintues) every 8 hours. After a minimum of 15 doses of IV therapy, patients who met
prespecified criteria of improvement could be switched to oral levofloxacin.1,2
In the treatment of cUTI
Don’t wait to defend your most vulnerable patients with VABOMERE2
Early appropriate treatment matters5
In an analysis of patients with CRE infections, those treated with an appropriate antibiotic within 48 hours of positive cultures experienced improved clinical and economic outcomes, including:
Clinical and real-world data support the early use of VABOMERE6,7
- In a randomized, prospective, open-label clinical trial, a subgroup of patients who received VABOMERE without prior antibiotic failure experienced improved clinical outcomes7
- In a retrospective, real-world study, patients with CRE infections including cUTI experienced fewer negative clinical outcomes when they started VABOMERE within 48 hours of culture collection.6
In the treatment of CRE cUTI, time matters.
Defend your appropriate patients with VABOMERE.5,6,8