Available susceptibility tests
VABOMERE® (meropenem and vaborbactam) is available for manual, automated, and semi-automated susceptibility testing.
The following susceptibility tests are approved for meropenem and vaborbactam:
Automated testing

NOW AVAILABLE
bioMérieux VITEK® 2 Gram-negative Susceptibility Panel
Ordering Information
NOW AVAILABLE
bioMérieux VITEK® 2 Gram-negative Susceptibility Panel
Ordering Information

- Meropenem/vaborbactam is available for the VITEK 2 systems (Note: Version 9 VITEK 2 software required)
- The VITEK 2 Gram-negative susceptibility panel is intended for use with the VITEK 2 systems in clinical laboratories as an in vitro test to determine the susceptibility of clinically significant aerobic Gram-negative bacilli to antimicrobial agents when used as instructed
- Refer to VITEK 2 AST panel package insert for FDA indications for use when testing meropenem/vaborbactam with VITEK 2 Systems
For more information, contact your local bioMérieux representative or visit go.biomerieux.com/vitek. To place and track orders online, visit biomerieuxDIRECT.com or call 1-800-682-2666.

Beckman Coulter MicroScan® MIC Panels
Ordering Information
Beckman Coulter MicroScan® MIC Panels
Ordering Information

- Meropenem/vaborbactam is available for MicroScan manual, autoSCAN®-4 and WalkAway® instrument systems testing
- The MicroScan MIC Panel contains 30 clinically relevant antimicrobial tests designed to reduce offline testing and improve lab workflow for multi-drug resistant organisms
For more information, contact your local Beckman Coulter Microbiology representative, visit beckmancoulter.com or to order call (800) 526-3821.

BD Phoenix™ Emerge™ gram-negative susceptibility panels from Becton Dickinson
Ordering Information
BD Phoenix™ Emerge™ gram-negative susceptibility panels from Becton Dickinson
Ordering Information
- The BD Emerge™, combination, and AST only panels are intended for use on the BD Phoenix™ identification and susceptibility testing instruments in clinical laboratories as an in vitro diagnostic test to determine the susceptibility to select aerobic gram-negative bacilli to antimicrobial agents as instructed by the BD Phoenix™ labeling
- For in vitro diagnostic use only. Observe approved biohazard precautions and aseptic techniques. This product is to be used only by adequately trained and qualified laboratory personnel. Sterilize all biohazard waste before disposal
- Laboratories will need to ensure the BD Phoenix™ instrument is configured to match the panel type. For additional questions, contact BD at 800-638-8663
For more information, contact your local BD Diagnostic Account Representative or visit
www.bd.com/en-us/offerings/capabilities/microbiology-solutions/identification-and-susceptibility-testing.
Manual and semi-automated testing

ETEST® available for automated testing from bioMérieux
Ordering Information
ETEST® available for automated testing from bioMérieux
Ordering Information
*Meropenem concentration 0.004-64 μg/mL-vaborbactam fixed concentration 8 μg/mL.
For more information, contact your local bioMérieux Inc. representative or call customer service at 800-682-2666 or visit www.biomerieux-usa.com/ETESTMEV.
To place an order, visit: www.biomerieuxDIRECT.com.
MIC Test Strip available from Liofilchem®
Ordering Information
MIC Test Strip available from Liofilchem®
Ordering Information

*Meropenem is a gradient in the MIC Strip: 0.016-256 μg/mL; vaborbactam is at fixed concentration along the strip.
- US customers can place an order by phone: 781-902-0312 or e-mail: orders@liofilchem.us.
- MTS strips are also available from Fisher Scientific: Catalog #22-777-842 (10/PK), #22-777-840 (30/PK), and #22-777-841 (100/PK).
For more information, contact liofilchem@liofilchem.net.
Susceptibility Disk from Hardy Diagnostics
Ordering Information
Susceptibility Disk from Hardy Diagnostics
Ordering Information

- Disks are clearly marked on both sides with letters and numbers designating the agent and the drug content
- The cartridges are for use in single-disk dispensers or multiplace dispensers and compatible with dispensers such as BBL™
To order, visit hardydiagnostics.com or call 1-800-266-2222.

Thermo Scientific™ Oxoid Disk and Sensititre™ MIC plates from Thermo Fisher Scientific
Ordering Information
Thermo Scientific™ Oxoid Disk and Sensititre™ MIC plates from Thermo Fisher Scientific
Ordering Information
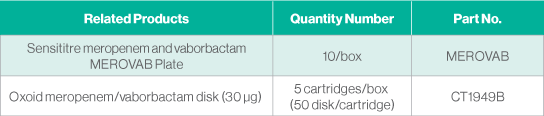
- Meropenem and vaborbactam is available for disk diffusion testing using Oxoid™ disks and on Sensititre™ panels for manual and fully automated testing
For more information, contact your local Thermo Fisher Scientific Microbiology representative at microbiology@thermofisher.com or visit thermofisher.com/AST.
FDA-recognized antimicrobial susceptibility test interpretive criteria for meropenem and vaborbactam
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC.
Ordering and billing