VABOMERE®—restoring the power of meropenem against CRE1
VABOMERE® (meropenem and vaborbactam) combines meropenem, a trusted carbapenem, with vaborbactam, a unique β-lactamase inhibitor (BLI)1

- Proven to work against a broad spectrum of gram-negative pathogens2,3
- Well-established efficacy and safety profile2,3

- First in a new class of cyclic boronic acid BLIs4
- Covalent bonding to target with extended resident time allowing meropenem to act unhindered5
VABOMERE addresses the challenge of resistant Enterobacterales5
WATCH THE VABOMERE MECHANISM OF ACTION
VABOMERE has a bactericidal effect on designated susceptible gram-negative organisms1
Vaborbactam1:
- Protects meropenem from degradation by a number of serine β-lactamases
- Does not have any antibacterial activity itself
- Does not decrease the activity of meropenem against meropenem-susceptible organisms
Meropenem1:
- Penetrates the cell wall of most gram-positive and gram-negative bacteria
- Binds with penicillin-binding protein (PBP) targets
- Initiates bacterial cell wall rupture and destruction
VABOMERE was specifically designed to be effective against key multidrug-resistant organisms, and has demonstrated no cross-resistance with other classes of antimicrobials.1,6
Meropenem and vaborbactam—designed to work together1
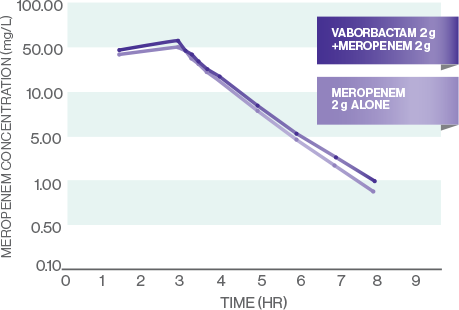
The pharmacokinetics (PK) of vaborbactam are well matched for meropenem1
MEROPENEM PK
VABORBACTAM PK
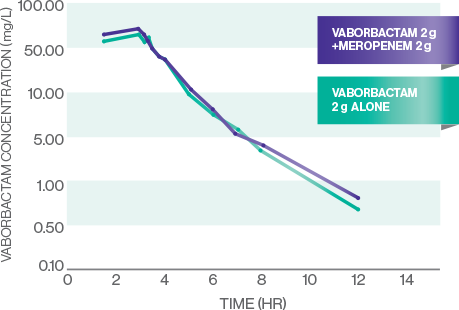
- The PK of meropenem and vaborbactam are linear across the dose range studied: 1 g-2 g for meropenem and 0.25 g-2 g for vaborbactam1
- All evaluated PK measures (Cmax, AUC0-t, and AUC0-inf) were similar for meropenem and vaborbactam alone and meropenem-vaborbactam in combination, indicating that concomitant administration of meropenem and vaborbactam did not affect the plasma PK of either drug7
- There were no plasma or urine PK drug-drug interactions7
The PK profiles of meropenem and vaborbactam are well matched, and the optimized dosing regimen of VABOMERE leads to target attainment against most commonly encountered CRE.5
VABOMERE dosing is optimized for strong bactericidal activity8-10
A 3-hour infusion provides optimized PK/PD exposures that lead to enhanced bacterial killing regardless of renal function8-11*†
PK/PD Target Attainment, Enterobacterales isolates11



PK/PD Target Attainment, KPC-Producing Enterobacterales isolates11



- The probability of target attainment exceeds 90% for susceptible MIC breakpoint (4 μg/mL) and intermediate breakpoint (8 μg/mL) in all renal function groups11
STUDY DESCRIPTION: Four simulated patient populations (n=1,000 each) varying by estimated glomerular filtration rate (eGFR) were generated using a uniform probability distribution for the following groups: ≥50, ≥30 to 49, ≥15 to 29, and <15 mL/min/1.73 m2. Using meropenem and vaborbactam population PK models, individual post hoc parameter estimates were generated. Using these estimates, total-drug concentration-time profiles were generated for simulated patients. An algorithm to assess PK/PD target attainment at meropenem and corresponding meropenem-vaborbactam MIC values based on worldwide in vitro surveillance data for Enterobacterales and KPC-producing Enterobacterales was followed.11
*In vitro activity does not necessarily correlate with clinical efficacy.
†Based upon meropenem-vaborbactam MIC distribution for 1,331 KPC-producing Enterobacterales isolates.11
MIC=minimum inhibitory concentration; PD=pharmacodynamic; PK=pharmacokinetic.